Bio-Hermes-002: A landmark study to revolutionise Alzheimer’s diagnosis
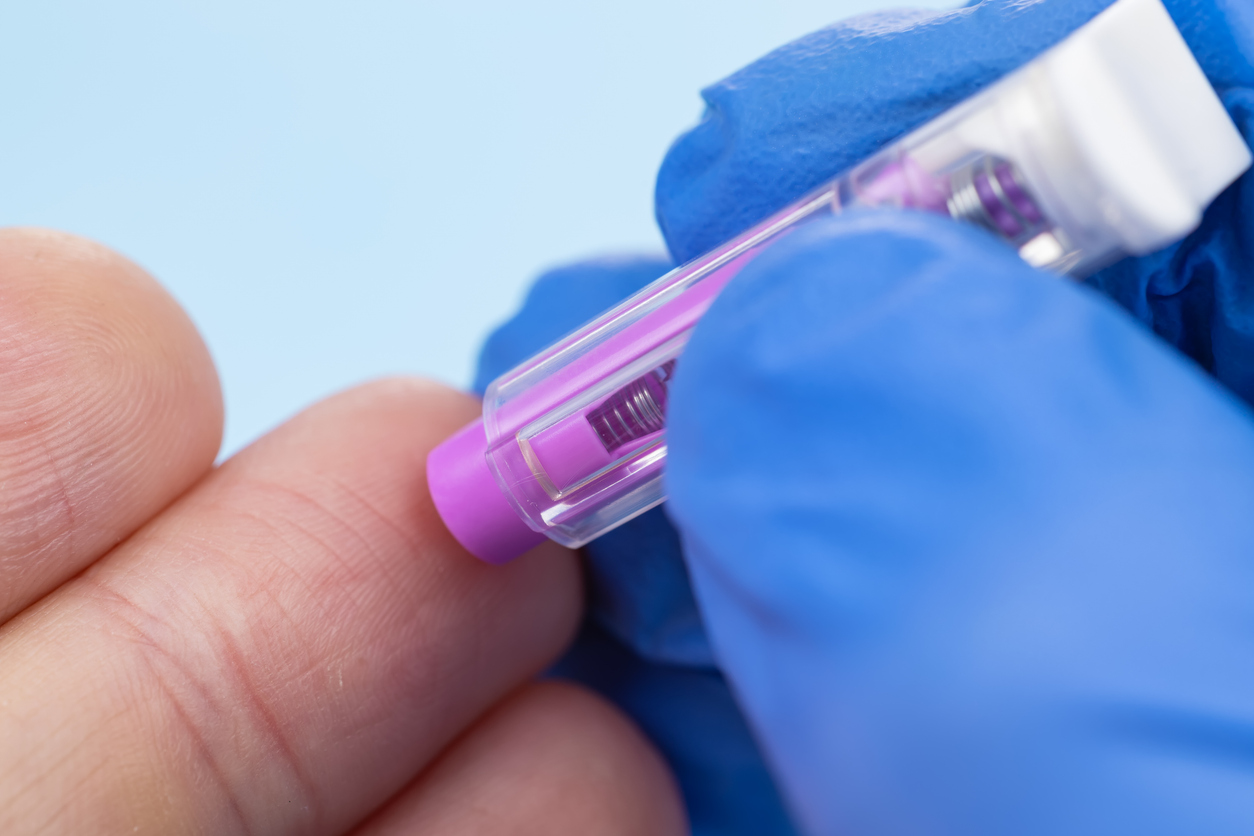
We’ve partnered with the Global Alzheimer’s Platform Foundation (GAP) to fund a trial of a finger prick-style test for Alzheimer’s disease as part of the Bio-Hermes-002 trial, with the support of the UK Dementia Research Institute (UKDRI).
Overview
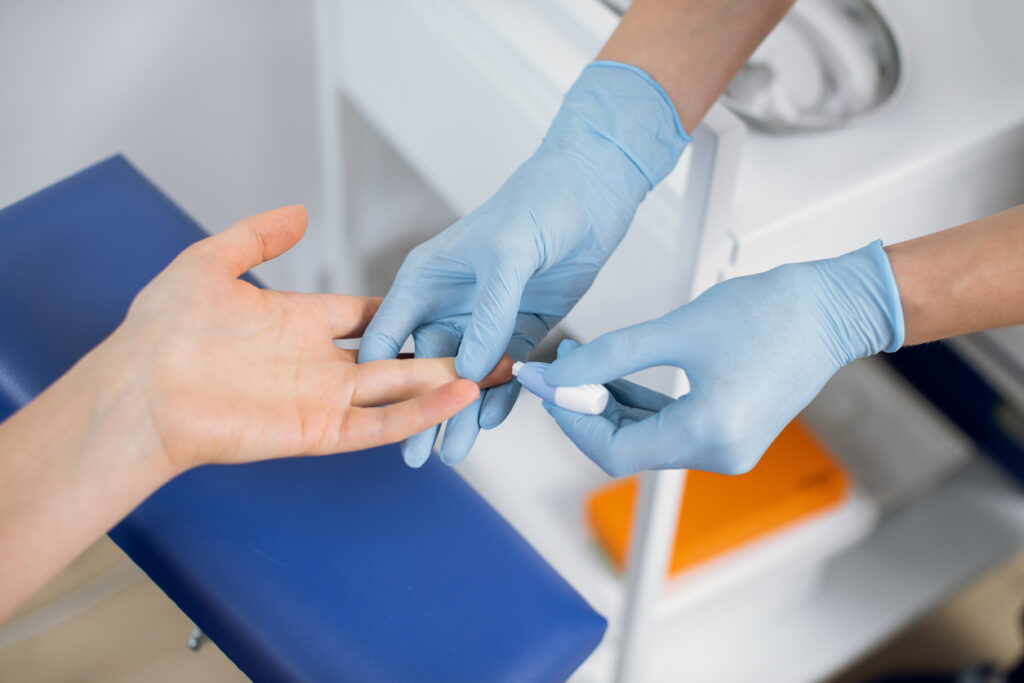
Our collaboration with GAP and UK DRI aims to transform Alzheimer’s diagnosis by validating a cost-effective, minimally invasive blood test. This international study will enrol 1,000 participants across the UK, USA and Canada, with at least 25% from underrepresented communities. By comparing blood-based biomarkers with current gold-standard methods, we hope to make early diagnosis accessible to all. This is the largest and most diverse international study testing whether this approach could help diagnose Alzheimer’s disease earlier and more affordably.
About Bio-Hermes-002
The study builds on the success of Bio-Hermes-001, the first-of-its kind study comparing blood-based biomarkers, digital cognitive tests, retinal exams and speech analytics to traditional methods of measuring cognition.
Now, as part of the Bio-Hermes-002 trial, we are exploring whether a finger-prick blood test can detect Alzheimer’s disease before symptoms appear. The test looks for three key proteins in the blood – phosphorylated tau 217 (pTau217), Glial fibrillary acidic protein (GFAP) and Neurofilament light polypeptide (NfL) – and compares results with the current gold-standard tests used to diagnose the disease – PET scans, MRI scans and lumbar punctures – and other emerging digital biomarkers. If successful, this approach could offer a scalable, accessible and affordable alternative to invasive procedures, enabling earlier intervention and better outcomes for patients.
About Alzheimer’s disease
Alzheimer’s disease is the most common cause of dementia, leading to progressive memory loss, confusion and cognitive decline. It affects millions worldwide and currently requires costly and invasive diagnostic methods such as brain scans and lumbar punctures. These barriers often delay diagnosis, leaving patients without timely access to treatments that could slow disease progression. A simple blood test could change this, making early detection possible for everyone, regardless of geography or healthcare access.

Earlier diagnosis of Alzheimer’s disease, through cheaper, scalable and more accessible tests is vital in the battle against this devastating condition. We are excited about the prospect of the finger-prick blood test because it will allow more patients to access new drugs, currently being developed, to slow disease progression in its early stages.”
Dr Giovanna Lalli, Director of Strategy and Operations, LifeArc
Contact us
In submitting your personal data via this form, you consent to being contacted via the details provided so that your enquiry can be responded to. If you would like your data to be removed, please email dataprivacy@lifearc.org.
Please see our Privacy Policy in relation to the personal data you submit to us through this page.